COVID Vaccine Study Halted: A Controversial CDC Move
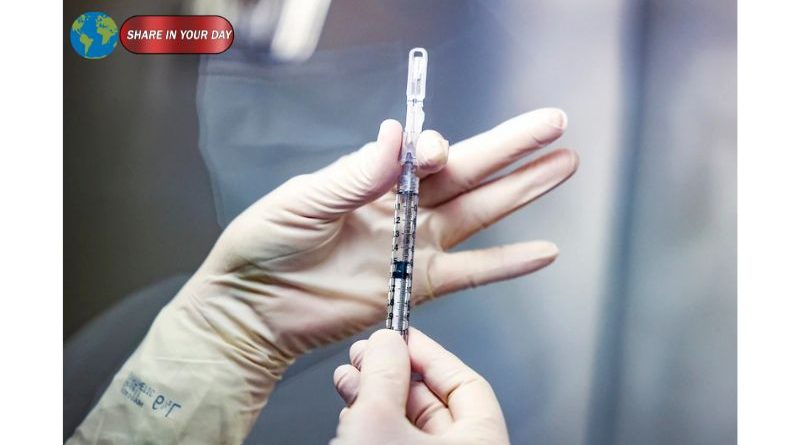
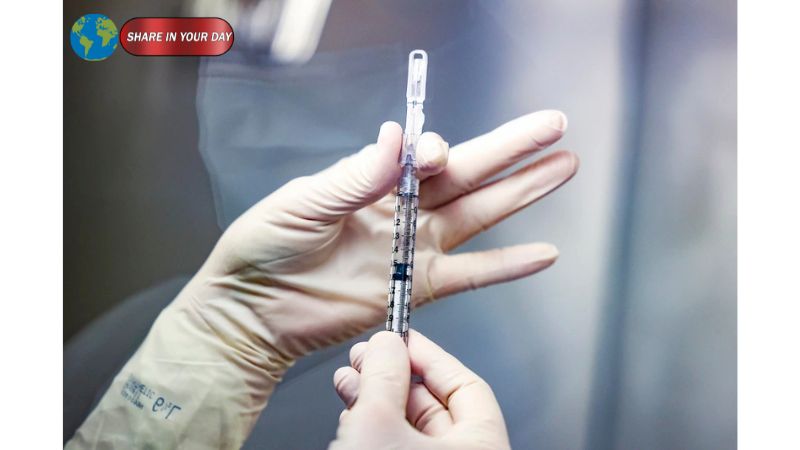
A covid vaccine study halted by the U.S. Centers for Disease Control and Prevention (CDC) has ignited widespread debate among scientists, policymakers, and public health experts. The study, which reportedly showed that COVID-19 vaccines significantly reduced hospitalizations and emergency visits, was stopped just before publication—raising questions about transparency and scientific independence.
The decision has become one of the most controversial public health developments of 2026, especially as it comes amid ongoing political tensions surrounding vaccine policy in the United States.
What the Halted Study Revealed
According to reports, the blocked study found that COVID-19 vaccines reduced the risk of emergency department visits and hospitalizations by roughly 50% among healthy adults during the most recent winter season.
This finding aligns with previous research suggesting that vaccination remains an effective tool in preventing severe illness, even as the pandemic has transitioned into a more manageable phase.
The study had already passed multiple levels of scientific review and was scheduled for publication in the CDC’s flagship journal, the Morbidity and Mortality Weekly Report (MMWR)—a key source of guidance for doctors and public health officials.
However, despite reaching this advanced stage, the publication was abruptly halted.
Why the CDC Blocked the Publication
Officials from the U.S. Department of Health and Human Services (HHS) stated that the study was not published due to concerns about its methodology. Specifically, they argued that the approach used to estimate vaccine effectiveness may not have met the agency’s standards.
The methodology in question compared outcomes between vaccinated and unvaccinated individuals who sought hospital care. Critics of the decision point out that this approach has been widely used in previous vaccine studies—including those published by the CDC itself.
This contradiction has fueled skepticism about whether the decision was purely scientific or influenced by external factors.
An Unusual and Rare Decision
Experts say it is highly unusual for a study that has already cleared internal review to be blocked at such a late stage.
Former CDC officials noted that they had rarely—if ever—seen a report halted after reaching the publication phase.
This has raised concerns about potential interference in the scientific process.
Some critics argue that delaying or suppressing data, especially data that demonstrates vaccine effectiveness, could undermine public trust in health institutions.
Political Context Behind the Decision
The controversy surrounding the covid vaccine study halted cannot be separated from the broader political environment.
The decision comes during a period of significant changes in U.S. vaccine policy under Health and Human Services Secretary Robert F. Kennedy Jr., who has long been known for skepticism toward vaccines.
Reports suggest that shifts in policy—such as moving away from broad vaccine recommendations toward individualized decision-making—have already altered how vaccine guidance is communicated.
At the same time, leadership instability within the CDC has added to the uncertainty, with key positions remaining unfilled and internal disagreements becoming more visible.
Scientific Community प्रतिक्रिया (Reaction)
The scientific community has responded with a mix of concern and criticism.
Some researchers argue that the halted study provided valuable real-world data that could help guide healthcare decisions, especially for vulnerable populations.
Others stress that rigorous methodology is essential and that delaying publication to ensure accuracy is not inherently problematic.
However, critics counter that the same methodological approach has been used in other peer-reviewed studies, making the justification for blocking this particular report questionable.
This divide highlights a broader tension between scientific caution and the need for timely information during ongoing public health challenges.
Impact on Public Trust
One of the biggest risks associated with the covid vaccine study halted is its potential impact on public trust.
Public health agencies rely heavily on credibility and transparency. When studies are delayed or withheld, it can create confusion and fuel misinformation.
Experts warn that even the perception of political influence can damage trust, regardless of whether the decision was scientifically justified.
In an era where misinformation spreads rapidly online, clear and consistent communication is more important than ever.
The Role of MMWR in Public Health
The CDC’s Morbidity and Mortality Weekly Report (MMWR) has long been considered one of the most authoritative sources of public health data in the United States.
Healthcare providers, researchers, and policymakers depend on it for timely and accurate information.
Blocking a study from appearing in this publication is not just an editorial decision—it has real-world implications for how medical professionals understand and respond to emerging health threats.
Broader Challenges in Vaccine Research
The halted study also reflects broader challenges facing COVID-19 vaccine research.
As the pandemic evolves, conducting large-scale studies has become more difficult due to declining case numbers, reduced public participation, and changing virus variants.
In fact, other vaccine-related studies have also faced setbacks, including halted clinical trials due to low enrollment.
These challenges make every piece of reliable data even more valuable.
What Happens Next?
It remains unclear whether the halted study will eventually be revised and published or permanently shelved.
Some experts are calling for greater transparency, including releasing the study data for independent review.
Others are urging policymakers to ensure that scientific decisions remain free from political influence.
The situation is still developing, and further updates are expected as more information becomes available.
Why This Matters Globally
Although the decision was made in the United States, its implications extend far beyond.
The CDC is one of the world’s leading public health institutions, and its actions often influence global health policies.
Countries around the world look to the CDC for guidance on vaccination strategies, pandemic response, and disease prevention.
Any controversy surrounding its decisions can have ripple effects across international health systems.
Conclusion
The covid vaccine study halted by the CDC represents a critical moment in the ongoing evolution of public health policy.
On one hand, ensuring scientific accuracy and methodological rigor is essential. On the other, transparency and timely communication are equally important in maintaining public trust.
As debates continue, the incident serves as a reminder that science does not operate in a vacuum—it is often shaped by political, social, and institutional forces.
Ultimately, how this situation is resolved may have lasting consequences for both public confidence in vaccines and the credibility of global health institutions.